The Legalization of Medical Cannabis in the European Union
Introduction
Medical Cannabis in the European Union is an important topic for cannabis enthusiasts. The landscape of medical cannabis legalization is evolving rapidly across the European Union (EU). With 21 out of 27 member countries having legalized medical cannabis, the region is experiencing significant shifts in healthcare, regulation, and market dynamics. This article explores the current state of medical cannabis legalization in the EU, the implications for various stakeholders, The future outlook for this burgeoning industry.
Furthermore,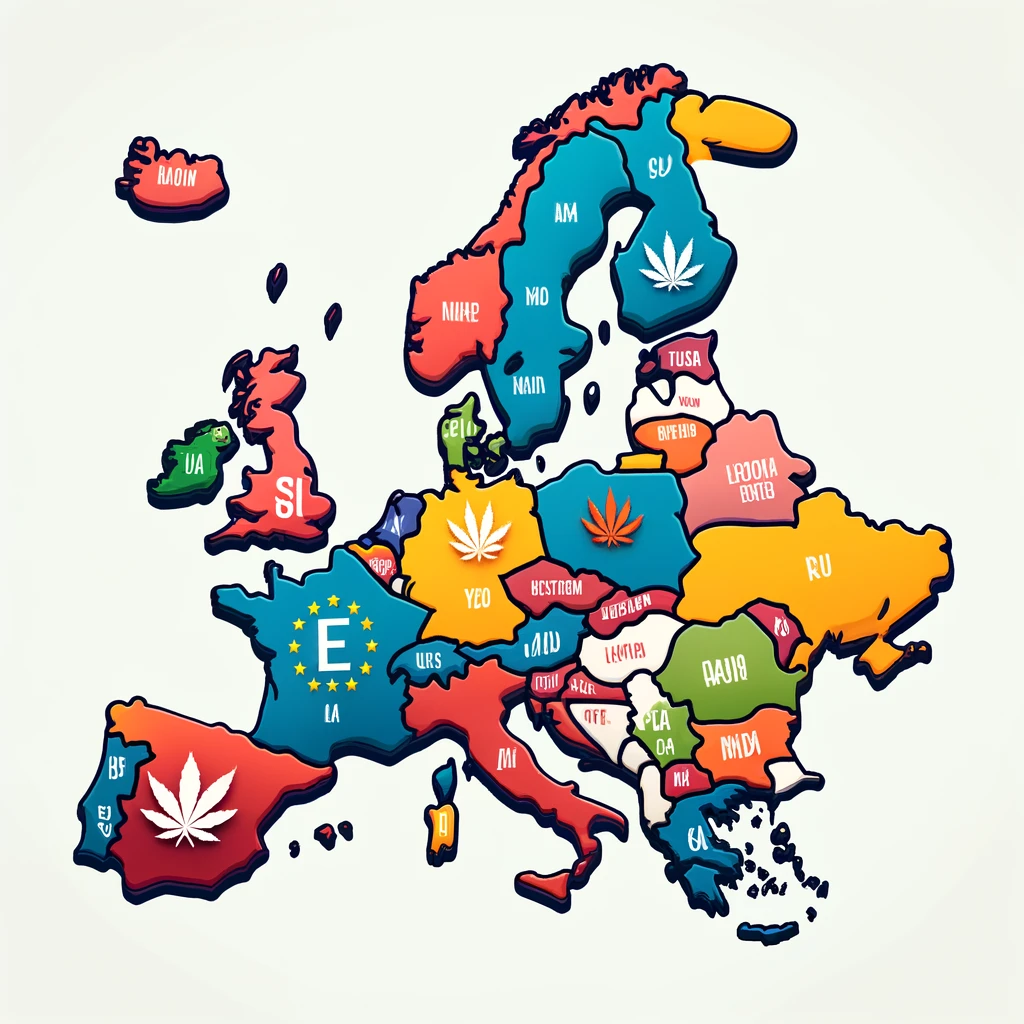
Overview of Legalization Status
Current Legal Framework
Moreover,What’s more, as of the latest reports, 21 EU countries have legalized medical cannabis to varying degrees. These countries include major markets such as Germany, Italy, The Netherlands, where comprehensive regulatory frameworks have been established to govern the production, distribution, and use of medical cannabis.
Country-Specific Regulations
Additionally,- Germany: Germany is one of the largest markets for medical cannabis in the EU. The country has implemented stringent regulations to ensure product quality and patient safety. German patients can access medical cannabis through prescriptions, and the costs are often covered by health insurance.
- Italy: Italy allows medical cannabis use under strict medical supervision. The Italian government has also initiated domestic cultivation to reduce dependence on imports.
- Netherlands: Known for its progressive stance on cannabis, the Netherlands has a well-established medical cannabis program. Dutch patients can access cannabis through pharmacies, and the country also exports medical cannabis to other EU countries.
Implications for Stakeholders
Patients and Healthcare Providers
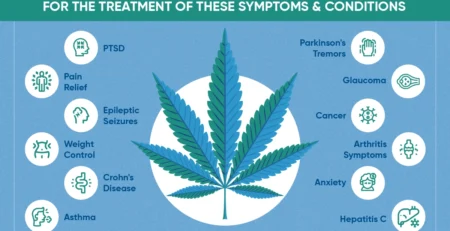
As a result,The legalization of medical cannabis has significant implications for patients and healthcare providers. Patients suffering from chronic pain, multiple sclerosis, epilepsy, Other conditions now have access to an alternative form of treatment that can improve their quality of life. Healthcare providers are increasingly incorporating medical cannabis into treatment plans, supported by growing evidence of its efficacy.
Pharmaceutical Industry
To put it simply, the pharmaceutical industry stands to benefit considerably from the legalization of medical cannabis. Companies are investing in research and development to create cannabis-based medicines that meet regulatory standards. The growing acceptance of medical cannabis also opens up opportunities for new product lines and revenue streams.
Regulatory Bodies
Regulatory bodies across the EU are tasked with ensuring that medical cannabis products meet safety and quality standards. This involves rigorous testing, certification processes, and ongoing monitoring to prevent misuse and ensure patient safety. The development of harmonized regulations across the EU could streamline these processes and facilitate the movement of medical cannabis products between member states.
Market Dynamics
Growth Potential
Interestingly, the European medical cannabis market is poised for substantial growth. According to industry analysts, the market could reach several billion euros within the next decade. Factors driving this growth include increasing patient acceptance, expanding legalization, and advancements in cannabis-based medicine.
Investment Opportunities
The burgeoning medical cannabis market presents lucrative investment opportunities. Venture capital firms, private equity, and institutional investors are increasingly interested in funding cannabis startups and established companies. Investments are being directed towards cultivation, processing, distribution, and research to capitalize on the market’s growth potential.
Challenges and Risks
Essentially, despite the positive outlook, the medical cannabis industry faces several challenges and risks. These include regulatory uncertainties, market fragmentation, and the need for extensive clinical research to validate the therapeutic benefits of cannabis. Companies must navigate these challenges to succeed in this competitive and evolving market.
Future Outlook

Harmonization of Regulations
One of the key trends to watch is the potential harmonization of medical cannabis regulations across the EU. A unified regulatory framework could simplify compliance for companies, facilitate cross-border trade, and ensure consistent product quality and safety standards. This would significantly benefit the industry and patients alike.
Advances in Research
Naturally, ongoing research into the medical applications of cannabis is expected to yield new insights and therapeutic products. Clinical trials and studies are essential to building a robust evidence base that can support the integration of cannabis into mainstream medicine. Advances in research will also drive innovation and the development of novel cannabis-based treatments.
Expansion of Access
Efforts are underway to expand access to medical cannabis for patients across the EU. This includes initiatives to increase the number of prescribing physicians, improve patient education, and reduce costs. As access improves, the demand for medical cannabis is likely to grow, further propelling the market.
Conclusion
Of course, the legalization of medical cannabis in 21 EU countries marks a significant milestone in the region’s approach to healthcare and regulation. The implications for patients, healthcare providers, the pharmaceutical industry, and regulatory bodies are profound. As the market continues to grow and evolve, stakeholders must navigate challenges and seize opportunities to ensure the successful integration of medical cannabis into the European healthcare landscape.
This comprehensive overview highlights the current state of medical cannabis legalization in the EU, offering insights into market dynamics, stakeholder implications, and future trends. By staying informed and adaptive, stakeholders can contribute to the responsible and effective use of medical cannabis in improving patient outcomes.
📚 Related Ottawa Cannabis Delivery Guides
🌿 Ready to Order?
Same-day delivery in Ottawa · 30-90 minutes · Free on orders $100+ · 613-614-2889